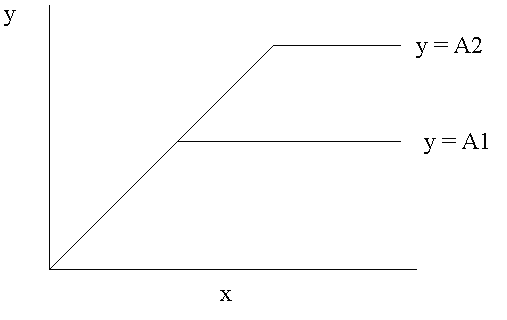
(Optimizing Nutrient Ratios in Mixed Fertilizers)
Copyright 2000 by Ernest M. Trionfo (All rights reserved.)
emt@trionfopublishing.com
When plants appear sickly due to a nutritional deficit the productivity and profits of the farming enterprise are likely to be low. The outlook may be turned around, however, by proper diagnosis of the plant and chemical analysis of the soil and the plant (Tisdale, 1975) followed by the application of the needed nutrient(s) in the form of an appropriate fertilizer. The fertilizer chosen depends on the composition of the soil and the nutritional status of the crop. It is desirable to have the same level of nutrient in the soil as is present in a fertile soil. Similarly, the elemental composition of the plant should be the same as in a healthy plant. A particular nutrient may be found to be lacking. If so, the addition of this nutrient should yield a growth response which is proportional to the amount of nutrient added.
Chemical analysis of the soil may reveal which nutrient is limiting plant growth but the question as to how much of this nutrient should be added depends on the particular plant species to be grown and the relative amounts of other nutrients present in the soil.
Liebig believed that plants use nutrients in a definite ratio. His "Law of the Minimum" predicts a strict relationship between the addition of a growth limiting nutrient and plant growth. He assumed that plant growth responds to increasing amounts of nutrient in a linear fashion in a manner analogous to an irreversible chemical reaction. This concept is illustrated in figure 1.
For example if you have 10 moles of oxygen gas and you add 1 mole of hydrogen gas and give it a spark the resulting explosion would give you 1 mole of water with 91/2 moles of oxygen gas left over.
1 H2 + 10 O2 --> H2O + 91/2 O2
If you used 2 moles of hydrogen gas instead of 1 this would give you 2 moles of water with 9 moles of oxygen gas left over.
2 H2 + 10 O2 --> 2 H2O + 9 O2
Three moles of hydrogen would give you 3 moles of water with 81/2 moles of oxygen gas left over.
3 H2 + 10 O2 --> 3 H2O + 81/2 O2
Four moles of hydrogen would give you 4 moles of water with 6 moles of oxygen gas left over.
4 H2 + 10 O2 --> 4 H2O + 6 O2
Here the limiting factor in the reaction is hydrogen and oxygen is in excess. A plot of the number of moles of hydrogen gas added vs. the yield in moles of water will be linear untill enough hydrogen is added to use up all of the oxygen at which point the plot will become horizontal.
The optimal ratio of hydrogen to oxygen is 2 to 1 thus when 20 moles of H2 are added to the 10 moles of O2 exactly 20 moles of water is produced with no oxygen left over.
20 H2 + 10 O2 --> 20 H2O
Below the optimal ratio hydrogen is the limiting factor for the reaction. Above the optimal ratio it is the oxygen which becomes the limiting factor. When 25 moles of H2 are added to the 10 moles of O2 exactly 20 moles of water is produced but this time with 5 moles of H2 in excess.
25 H2 + 10 O2 --> 20 H2O + 5 H2
The production of more water is possible only when the amount of oxygen is increased.
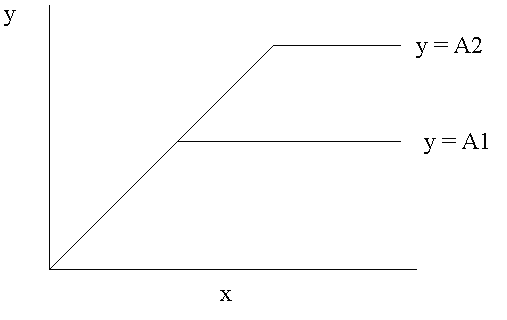
Figure 1. Liebig's Law of the Minimum. The nutrient in the minimum refers to the nutrient in the least relative quantity in the soil. It is the growth limiting nutrient. All other nutrients are in excess. Liebig's Law predicts that when the nutrient in the minimum (X) is added in increasing amounts to a plant correspondingly increasing yields (Y) are obtained untill a second nutrient becomes in the minimum. A plot of the amount of X added vs. Y will be linear untill the second nutrient becomes limiting. At this point the yield curve will be flat as indicated by the horizontal line y = A1 which is the maximum yield possible under the given conditions. According to the law when a second growth limiting nutrient is increased a higher maximum yield is possible as indicated by the line y = A2.
Thus, according to Liebig the yield increase of the crop (Y) is assumed to be directly proportional to the increase of a growth limiting nutrient (X) or
dY = KdX
untill the maximum yield (A) is achieved. Where K is a constant such that
K = dY/dX
MITSCHERLICH'S LAW OF PHYSIOLOGICAL RELATIONS
Mitscherlich (1909) , however, found that the yield response of plants to an increase of a limiting nutrient was proportional to the decrement from the maximum yield attainable (A):
dY/dX = (A - Y) K.
Where both A and K are constants.
On integration this differential equation yields
Ln (A - Y) = C - KX
Where C is a constant.
The exponential form of the equation is:
Y = A(1 - e-kx) or Y = A(1 - e-(ln2/h) x)
Where h = the quantity of nutrient required to give a half maximum yield also known as a "Baule unit".
Baule (1918) suggested that Mitscherlich's law was a contradiction of Liebig's Law in that unlike the Law of the Minimum each nutrient promotes growth in a completely independent manner. Baule noticed that application of a fertilizer to 3 plots of differing fertility increased the yield of each plot by the same percentage. One half of each plot was fertilized and the other half not fertilized. In each case fertilization doubled the yield regardless of the degree of fertility of the field.
Baule predicted that the addition of a compound fertilizer would increase the fractional yield of a crop according to the following equation:
Y/A = (1 - e-x1(ln2/h1))(1 - e-x2(ln2/h2))(1 - e-x3(ln2/h3)) . . .(1 - e-xn(ln2/hn) )
Where h1 is the quantity of nutrient required to give a half maximum yield for the 1st nutrient X1, h2 is the quantity of nutrient required to give a half maximum yield for the 2nd nutrient X2 and so on. Thus, the total yield would be
Y = A (1 - e-x1(ln2/h1))(1 - e-x2(ln2/h2))(1 - e-x3(ln2/h3)) . . .(1 - e-xn(ln2/hn) )
The above equation represents Baule's "general effect law". Baule's general effect law tells us that although the application of a compound fertilizer (e.g. phosphorus + nitrogen + potassium ) will always result in a greater crop yield--- the percentage increase in the yield per quantity of fertilizer added will always be less than the percentage increase in yield per quantity of fertilizer added for a simple fertilizer (just phosphorus, just nitrogen or just potassium).
Another way of stating Baule's general effect law is that for each nutrient the quantity of nutrient required to produce half the maximal yield (h) is invariant regardless of the relative quantity of any other nutrient. The validity of Baule's general effect law, then, is dependent on the invariance of the h values of nutrients.
In reviewing the literature DeVries (1939) cast doubt on the invariance of h. He suggested that the shape of the nutrient growth response curve depends upon how nearly in the optimum other nutrients are. In other words the use efficiency of nutrients are interdependent. This concept is known as Liebscher's Law of the Optimum.
Liebscher's Law of the Optimum predicts an increase in the use efficiency of nutrients as other nutrients are brought closer to the optimum. Liebscher's Law was originally described as of a modification of Liebig's Law of the Minimum as illustrated in figures 2 and 3.
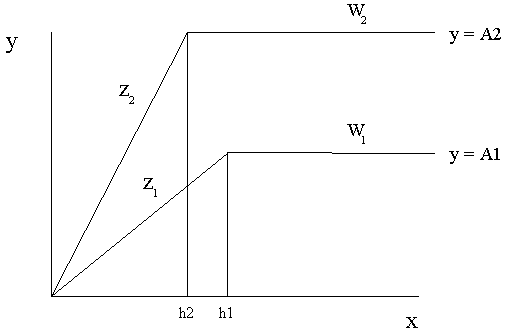
Figure 2. Liebscher's Law of the Optimum predicts an increase in the use efficiency of the nutrient X as a second nutrient W is brought closer to the optimum. The use efficiency of X is determined by finding the slope Z of the line of the minimum (where Z 2> Z 1 ). Y represents the yield. Two different levels of the 2nd nutrient (W1 and W2) are shown. A1 and A2 represent maximum yields for the amounts of the 2nd nutrient W1 and W2 respectively. Thus the use efficiency of nutrient X is dependent on the quantity of nutrient W. In actuality the growth response is curvilinear and the lines with the slopes Z 2 and Z 1 represent oblique asymptotes while the lines W2 and W1 represent horizontal asymptotes. The values h1 and h2 represent the amounts of nutrient X which are required to produce a half maximal growth response to X at the levels of W1 and W2 respectively.
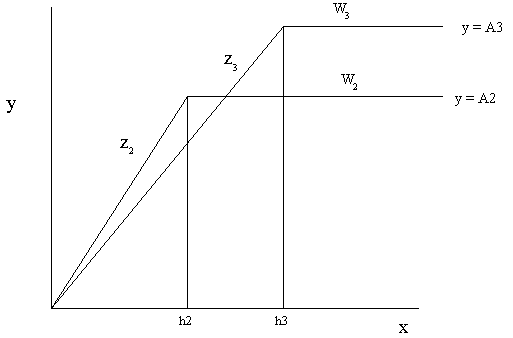
Figure 3. When the amount of the nutrient W is increased above the optimum the use efficiency of X decreases to Z3 and the half maximal growth response of to X is increases to h3.
As we can see from these graphs as the use efficiency of X (represented by the slope Z) is increased from Z1 to Z2 by increasing nutrient W from W1 to W2. The value of h initially decreases from h1 to h2 but as the amount of W is increased above the optimum the use efficiency decreases from Z2 to Z3 and the value of h increases from h2 to h3.
We shall examine the data of DeVries on the effect of adding increasing amounts of potash at various levels of ammonium nitrate on the growth of Wiesenchwingel (a type of grass) in more detail shortly.
Macy (1935) studied the yield of crops not in terms of the amount of nutrient added but the amount of nutrient incorporated into the plant. His chemical analysis of crops revealed three nutritional zones in the growth curve (figures 4 and 5):
The nutritional zones in growth curve are: 1. An area of where a constant minimum percentage of nutrient required for growth is present in the plant. 2. An area of increasing nutrient content which Macy describes as "poverty adjustment". 3. An area of increasing nutrient content which Macy describes as "luxury consumption".
The critical percentage is that percentage of nutrient in the plant above which vegetative growth is enhanced while blossoming and flowering are inhibited. It is found an the hypothetical point of inflection between the zones of "poverty adjustment" and "luxury consumption". These zones are shown in figure 4.
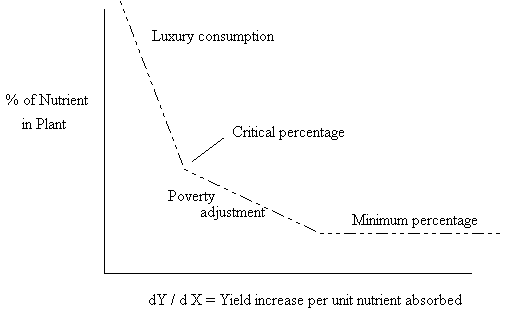
Figure 4. Macy's nutritional zones of plant composition.
Macy suggested that the Law of the Minimum was valid in the zone of minimum percentage and in the zone of luxury consumption while Mitscherlich's law was valid in the zone of poverty adjustment. Macy's nutritional zones with respect to the yield curve are shown in figure 5.
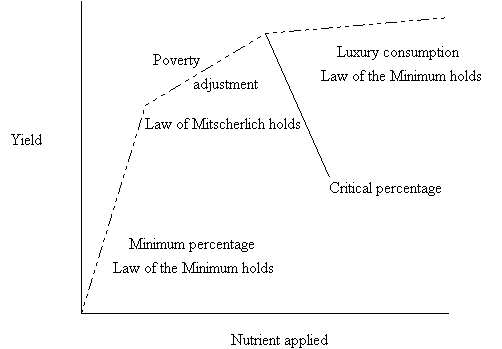
Figure 5. Macy's nutritional zones of plant composition as applied to the yield curve.
Boss(1962), however, showed that the Mitscherlich equation could adequately describe the growth of plants when weight is plotted as a function of nutrient absorption. Boss grew 1mm corn root tip sections in tissue culture in media containing incremental amounts of nitrogen and phosphorus. His analysis of the media after growth had stopped showed that all of the nitrogen and phosphorus had been absorbed. The data adequately fitted the Mitscherlich equation. His analysis, nevertheless, confirmed the important findings of Macy:
Nutrients which are present in excess of the plant's growth requirements are absorbed but do not contribute to plant growth. This excess absorption is called "luxury consumption".
THE RELATIVE USE EFFICIENCY OF NUTRIENTS
In order to unite these various ideas of plant growth we shall first consider some theoretical implications of the law of the minimum. We shall see that Liebig's law holds only for situations where growth conditions are ideal and when all nutrients except the one in the minimum are in essentially unlimited supply. Next we shall consider how the growth trajectories of plants are affected by 2 limiting nutrients. Finally, we will explain the law of the optimum in terms of the relative concentration and amounts of 2 nutrients.
Liebig conceived of plant growth in terms of an irreversible chemical reaction. But all reactions which occur in living cells take place in equilibrium and are catalyzed by enzymes. Growth occurs not continuously but in jumps or steps with each cellular division. Before each cell makes a commitment to divide all of the requirements for cellular division must be met. An excess of any nutrient has a feedback effect on the rate of cellular division. An effect which we shall discuss when we reconsider Liebig's Law of the optimum.
Now we shall perform a thought experiment. Let us consider the ideal situation as shown in figure 6. We will grow a plant capable of attaining infinite size. All nutrients except the one in the minimum (i.e. nutrient A) will be in unlimited supply so that no other nutrient becomes limiting to growth as we increase the amount of nutrient A.
The Law of the Minimum predicts a linear relationship between the growth of the plant and the quantity of nutrient A. Thus, increasing additions of nutrient A will give a straight line plot of nutrient A vs. the yield or Y. This line can be described by the equation Y = E Xa. Where E is the slope of the line and Xa is the amount of nutrient A.
If instead of adding nutrient A we add a nutrient which is used less efficiently (e.g. nutrient B) we will obtain a plot of nutrient B vs. the yield in which the line has a slope which is less steep than for nutrient A. This line can be described by the equation Y = G Xb. Where G is the slope of the line and Xb is the amount of nutrient B.
Now, if we draw a line horizontal to the X axis at any point along the Y axis we can obtain the maximum yield line as predicted by the Law of the Minimum. But the Y coordinate of the point of intersection of this line with the lines Y = E Xa and Y = G Xb are the same.
Now, we drop down 2 vertical lines between the intersection points of the maximum yield line and the lines Y = E Xa and Y = G Xb to the X axis. The points at which these lines intersect the X axis represent the points Ha and Hb respectively. Ha is the amount of nutrient A required to give a half maximal growth response to nutrient A when nutrient B is held constant at Hb and all other nutrients are in unlimited supply. Hb is the amount of nutrient B required to give a half maximal growth response to nutrient B when nutrient A is held constant at Ha and all other nutrients are in unlimited supply.
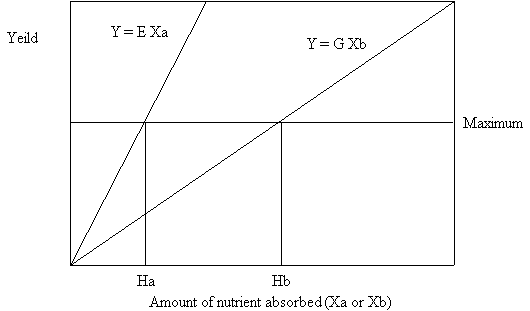
Figure 6. Relative use efficiency of two nutrients. Lines indicating the horizontal and oblique asymptotes of the hypothetical growth curves are shown. The vertical lines labeled Ha and Hb represent the half maximal inflection points of the growth response curves for nutrients A and B respectively.
If we increase the quantity of nutrient A the growth response of the plant will be curvilinear, with A limiting growth and B in excess from 0 to Ha. From Ha to infinity B will be limiting growth and A will be in excess. At the quantity Ha neither nutrient A nor nutrient B will be limiting growth and the ratio of the amount of nutrient A divided by the amount of nutrient B will be at the most use efficient ratio (Ha/Hb) possible for the given quantity of nutrient B. The asymptotes of the curve are shown diagrammatically in figure 7.
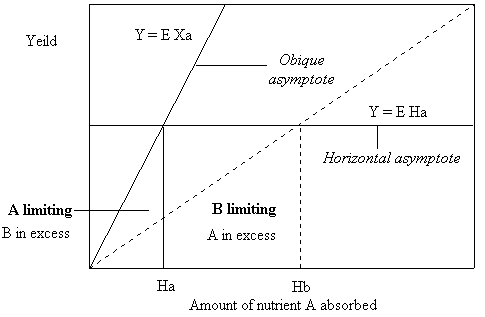
Figure 7. Growth curve asymptotes for the case where nutrient B is held constant at Hb and nutrient A is given to the plant in increasing amounts.
If, on the other hand, we increase the quantity of nutrient B the growth response of the plant will be curvilinear, with A limiting growth and B in excess from 0 to Hb. From Hb to infinity A will be limiting growth and B will be in excess. At the quantity Hb neither nutrient B nor nutrient A will be limiting growth and the ratio of the amount of nutrient B divided by the amount of nutrient A will be at the most use efficient ratio (Hb/Ha) possible for the given quantity of nutrient A. The asymptotes of this curve are shown diagrammatically in figure 8.
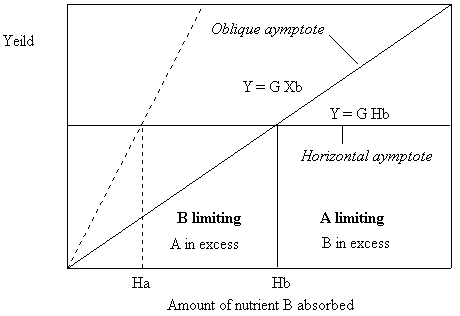
Figure 8. Growth curve asymptotes for the case where nutrient A is held constant at Ha and nutrient B is given to the plant in increasing amounts.
In the first case a plot which of the growth response of a plant to increasing amounts of nutrient A will have an oblique asymptote equal to Y = E A and a horizontal asymptote of Y = G Ha. In the second case, a plot which of the growth response of a plant to increasing amounts of nutrient B should have an oblique asymptote equal to Y = G B and a horizontal asymptote of Y = G Hb. Now, since Y = EHa = GHb we can derive the equations:
EHa = GHb,
E/G = Hb/Ha,
Ha = (G/E)Hb,
and
Hb = (E/G)Ha
The absorption of a non-limiting nutrient (say nutrient A) by dividing cells can be illustrated diagrammatically as follows:
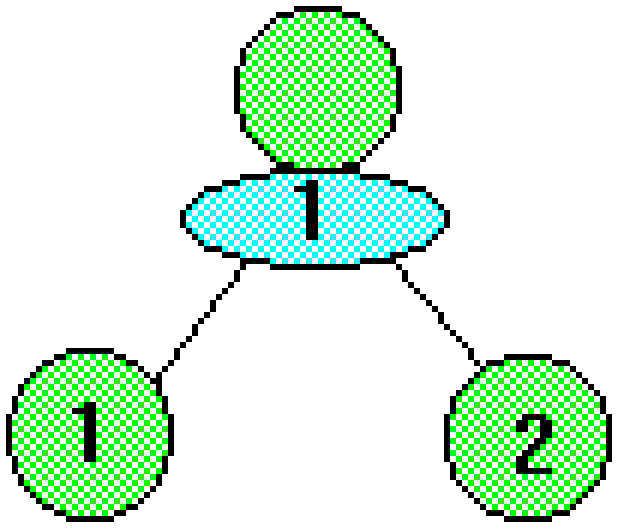
Dividing cells are represented as circles. Quantities of the growth limiting nutrient are represented as ovals.
During the 1st generation 2 cells are fored after the absorption of 1 unit of nutrient.
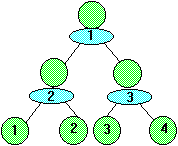
During the 2nd generation 4 cells are formed after the absorption of 3 units of nutrient.
During the 3rd generation 8 cells are formed after the absorption of 7 units of nutrient.
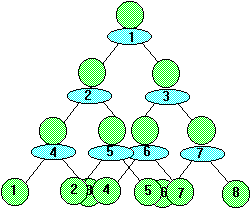
The increase in the number of living cells is directly proportional to the amount of nutrient A absorbed which follows the numerical series 1, 3, 7, 15... 2n -1. The actual number of cells produced follows the numerical series 1, 2, 4, 8, 16 ... 2n.
But when the plant has only a fixed quantity of nutrient B, greater and greater amounts of nutrient A are required for plant cells to divide. The more cells produced the more of nutrient A which is required for division. Thus, the increase of plant weight with increasing amounts of nutrient A is directly proportional to the numerical series 1, 3, 7, 15... 2n -1 and inversely proportional to the number of living cells produced which follows the numerical series 1, 2, 4, 8, 16 ... 2n.
Rauterberg (1939) has shown that the logarithmic form of Mitscherlich's equation is obtainable by substituting x / h for n in the equation
Y(n) / A = (2n-1)/2n
Where Y is the yield, A is the maxium yield and n is a mulitple of the quantity of nutrient required to give a half maximum yield (h), and n = X / h.
Y/A = (2(x / h) -1) / 2(x / h)
Y = A (2(x / h) -1) / 2(x / h)
Y = (A 2(x / h) - A) / 2(x / h)
Y = A - A /2(x / h)
Y - A = - A / 2(x / h)
A - Y = A / 2 (x / h)
ln (A - Y) = ln (A / 2 (x / h))
ln (A - Y) = ln (A) - ln (2 (x / h))
ln (A - Y) = ln (A) - (x / h) ln (2)
ln (A - Y) = ln (A) - (ln (2) / h) x
ln (A - Y) = c - k x
Mitscherlich's differential equation
dY/dX = (A - Y) K.
states that the growth increment is proportional to the decrement from the maximum yield obtainable.
Now let us consider the growth response of a plant to nutrient A such that the logarithmic form of the Mitscherlich equation is
ln (M - Y) = c - k A
and the exponential form is
Y = A (1 - e-(ln2 / Ha) Xa)
If we let A = E (Ha/ln2) then
Y(Xa) = E (Ha /ln2 ) (1 - e-(ln2 / Ha) Xa )
Similarly, if we consider the growth response of a plant to nutrient B such that the logarithmic form of the Mitscherlich equation is
ln (B - Y) = c - k B
and the exponential form is
Y = B (1 - e-(ln2 / Hb) Xb)
and if we let B = G (Hb/ln2) then we have
Y(Xb) = G (Hb /ln2 ) (1 - e-(ln2/Hb) Xb )
Now from the previous section we have concluded that
Ha = (G / E) Hb and
Hb = (E / G) Ha
Thus we have a means of comparing the growth response of a plant to one nutrient as a function of a second nutrient in the relativistic equations:
Y(Xa) = E (G/E) Hb /ln2 ) (1 - e-(ln2 /(G / E) Hb) Xa)
and
Y(Xb) = G( E/G) Ha /ln2 ) (1 - e-(ln2 / (E/G) Ha) Xb)
Which, of course, can be reduced to
Y(Xa) = (G Hb /ln2 ) (1 - e-(ln2 /(G / E) Hb) Xa) and
Y(Xb) = (E Ha /ln2 ) (1 - e-(ln2 / (E / G) Ha) Xb)
In order to illustrate how useful these equations are in determining the relative nutrient requirements of plants we shall first examine a hypothetical situation concerning the effects of various levels of nutrients A at different levels of nutrient B (and vice versa) on the shape of the growth of a hypothetical plant. Next we shall see how curves generated by these equations can be adequately fitted to the data of Devries (1939, after S. Landeck) concerning the effect of increasing quantities of potash at different levels of ammonium nitrate on the growth of Wiesenchwingel .
Figure 9 shows the growth response of a hypothetical plant to increasing quantities of nutrient A for various amounts of nutrient B according to the equation Y(Xa) = (E Ha)/ln2 (1- e -(ln2/Ha)Xa). Ha represents both the amount of nutrient A required to give a half maximal growth response to increasing quantities of nutrient A. It can be seen as the amount of nutrient B increases, the use efficiency of nutrient A increases and the curve approaches the oblique asymptote or Liebig's line of the minimum.
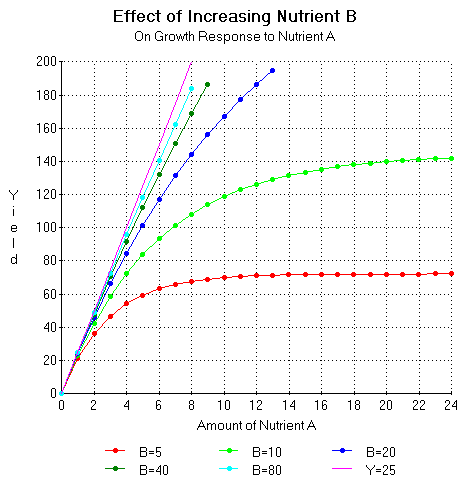
Figure 9. Growth response to increasing quantities of nutrient A for various amounts of nutrient B according to the equation Y(Xa) = (E Ha)/ln2 (1- e -(ln2/Ha)Xa). Ha represents the amount of nutrient A required to give a half maximal growth response to increasing quantities of A. Ha = (G/E) Hb. G = 10 and E = 25.
Figure 10 shows the growth response of a hypothetical plant to increasing quantities of nutrient B for various levels of nutrient A according to the equation Y(Xb) = (G Hb)/ln2 (1- e -(ln2/Hb)Xb). Hb represents the amount of nutrient B required to give a half maximal growth response to increasing quantities of nutrient B. Again, in this case as the amount of nutrient A is increased the use efficiency of nutrient B increases. Also, again, as the amount of nutrient A is increased the curve approaches the oblique asymptote or Liebig's line of the minimum.
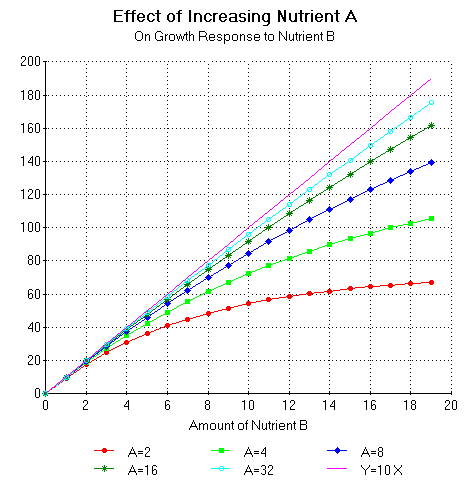
Figure 10. Growth response to increasing quantities of nutrient B for various amounts of nutrient A according to the equation Y(Xb) = (G Hb)/ln2 (1- e -(ln2/Hb)Xb). Hb represents the amount of nutrient B required to give a half maximal growth response to increasing quantities of B. Hb can be calculated from the formula Hb = (E/G) Ha, where G = 10 and E = 25.
To illustrate how these equations can be used to determine the most use efficient nutrient ratio of fertilizers, a computerized multiple regression analysis of the data of DeVries (1939) on the growth response of Wiesenchwingel to increasing levels of potash for different levels of ammonium nitrate is shown in figure 11 (click the image to see a demonstration of the computer program used). The experiment was conducted by S. Landeck.
The form on the right of the demo shows the growth response of Wiesenchwingel to potash at various levels of ammonium nitrate. The growth response of Wiesenchwingel to ammonium nitrate at various levels of potash is shown on the left of the demo.
The E asymptote was found to be 89.75 grams of plant tissue / gram of pottash (Nutrient A). The G asymptote was found to be 43.21 grams of plant tissue / gram of ammonium nitrate (Nutrient B). Therefore, the optimal use efficiency of ammonium nitrate to pottash by Wiesenchwingel is 2.08 (the ratio of E/G = Hb/Ha).
Figure 11. Growth of Wiesenchwingel with increasing quantities of potash at different levels of nitrogen fitted to relative nutrient growth response equations. Click image to see demonstration in an active-x document (requires Microsoft Internet Explorer). The program will load into your computer's memory but will not write to your disk.
The most use-efficient mixed fertilizer for a plant would be one that has optimally balanced nutrient ratios. For example, consider just the 3 major nutrients; nitrogen, potassium and phosphorus (N, K and P). Suppose that when we vary N with respect to K, when K is 80 grams and half maximal growth occurs at 40 grams of N then Hk = 80 grams the optimal ratio of N to K is 40:80 or 1:2. When N and K are held constant at 120 grams (40 grams of N plus 80 grams of K) and a half maximal yield occurs at 50 grams of P then the optimal ratio of NKP is 40:80:50 or 4:8:5.
If all of the essential nutrients of a particular plant were optimized in a mixed fertilizer in this fashion then we should expect that its use efficiency would be maximal with no single nutrient in excess and no single nutrient in the minimum.
Formulas which express a proportional relationship between different parts of a growing plant are called allometric or scaling formulas (Niklas, 1994). In allometric growth, one part of an organism is correlated with the growth of another part and the growth of both parts are functionally related to the growth of the whole organism (Laird, Anna Kane, 1965 and Laird, Anna Kane, et al.,1965.
Toughton (1955), for instance, found that in grasses the allometric relationship between shoot growth and root growth was affected by nitrogenous fertilizer which both increased the size of the plants and decreased the root growth relative to the shoot growth. The relationship he found was
Y1 = AY2B
which may be converted to a linear equation as follows:
lnY1 = ln AY2B lnY1 = ln A + lnY2B lnY1 = B lnY2 + ln A or
lnY1 = B lnY2 + A'
where, Y1 = root weight and Y2 = shoot weight, and A' and B are constants.
Laird (1965 ) has shown allometric relationships can be mathematically derived from a description of growth in terms of the Compertz growth equation. We can describe root and shoot growth using the following Compertzian formulas:
lnYr(t) = R (Hr / ln2)(1-e- (ln2/Hr) t ) and lnYs(t) = S (Hs / ln2)(1-e- (ln2/Hs) t )
Where Yr(t) is the growth of the root and Ys(t) the growth of the shoot at time t. The constants Hr and Hs are the times at which root and shoot growth, respectively, are half maximal. R and S are constants.
In the discussion which follows we shall make the simplifying assumption that the plant top or shoot constitutes the entire plant so that in this particular case
Yp(t) = EXP [S (Hs / ln2)(1-e- (ln2/Hs) t )]
This Compertzian relationship will be redefined, however, to express the constraining relationship to increasing amounts and concentrations of 2 different nutrients on the extent and rate of growth of the plant top.
Rauterberg's Hyperbolic Equation
Allometric plant growth as represented by the Compertzian growth equation eventually reaches a maximum which may occur at different times depending on the concentration of nutrients in the soil. When plants are harvested at the same time they may be at different points on their respective growth trajectories. The proper application of the Mitscherlich equation in the determination of the nutrient requirements of plants must take into account evidence that growth has stopped.
In order to demonstrate the importance of this concept we shall consider a hypothetical situation where plants are given various amounts of a limiting nutrient and then allowed to grow untill all of the limiting nutrient has been absorbed by the plant. But first we shall have to consider the effect of nutrient concentration on the growth rate of plants.
Rauterberg (1939) found that the yield curve under certain circumstances could be adequately described by the hyperbolic equation
Y = Ax / (h + x)
As discussed before Rauterberg showed that Mitscherlich's equation is obtainable by expressing the amount of nutrient added (x) in terms of a multiple (n) of the quantity of nutrient (h) required to give a half maximum yield and substituting this value into the equation (2n - 1) / 2n
That is by letting x = nh and substituting n = x / h into (2n - 1) / 2n we can obtain ln (A - Y) = c - k x.
Rauterberg, instead of substituting the factor x / h = n into the equation (2n - 1) / 2n substituted x / h = n into the equation n /(n+1) and obtained
n / ( n + 1 ) = ( x / h ) / [ (x / h) +1 ]
n / ( n + 1 ) = x / (x + h)
Y = [x / (x + h)] A
Y = xA / (x + h )
The above equation is the formula for a hyperbola which goes through the 0 point of a coordinate axis in which the principle axis is rotated by 90 degrees. It should also be recognized as the formalistic equivalent of the Michaelis-Menton equation in enzyme kinetic theory where Y expresses reaction rate, A expresses Maximum reaction rate and h is the Michaelis constant equivalent to the substrate concentration (x) required to give a half maximal reaction rate (h). Here, however, Y represents crop yield, A represents maximum crop yield, and h represents the quantity of nutrient (x) required to give a half maximal growth yield (h).
Table 1 shows a comparison between the Logarithmic equation of Mitscherlich and the hyperbolic equation of Rauterberg showing the relationship between the number of "effect quantities" added and the predicted fraction of the maximal yield obtainable by adding the given number of effect quantities. The effect quantity (nh) is a multiple of the amount of nutrient required to give a half maximal yield)
Table 1
Fraction of Maximum Yield
|
Effect Quantities |
Logarithmic Equation |
Hyperbolic Equation |
|---|---|---|
|
1h |
1/2 |
1/2 |
|
2h |
3/4 |
2/3 |
|
3h |
7/8 |
3/4 |
|
4h |
15/16 |
4/5 |
|
5h |
31/32 |
5/6 |
|
6h |
63/64 |
6/7 |
|
. |
. |
. |
|
. |
. |
. |
|
. |
. |
. |
|
nh |
(2n-1)/2n |
n/(n+1) |
The growth rate of human cells in tissue culture under optimal conditions responds hyperbolically to increasing nutrient concentrations according to the formula
dD/dt = A [x] / ([x] + B)
McKeehan et al. (1980) and McKeehan and McKeehan (1981).
Where D = Ln(Y), Y is the number of cells, and dD/dt = the rate of cell doublings per day at nutrient concentration [x]. The constant A represents the maximum growth rate and B is the nutrient concentration which supports a half maximal growth rate.
The units of concentration in this formula can be converted to amounts by multiplying the top and bottom of the equation by 1/V
dD/dt = A x / (x / (x + B'))
If we substitute Y(x) for doubling rate then this formula becomes
Y(x) = A x / (x / (x + B'))
which is the Rauterburg equation.
The logical conclusion is that in the situations where Rauterberg (1939) found that the yield curve could be better represented by the hyperbolic equation, growth had not stopped and all of the added fertilizer had not been absorbed by the plant. In essence what Rauterberg (1939) had done was measure the growth rate response of plants to increasing nutrient concentrations. This information gives us a way to describe the growth trajectories of plants in terms of both nutrient amount and concentration.
The maxima of plant growth can be describe by the Mitscherlich's equation while the growth rate or time require to reach a half maximum plant size can be describe by the Rauterberg equation. Thus, according to the Compertz equation
Yp(t) = EXP [S (Hs / ln2)(1-e- (ln2/Hs) t )]
or simply
Y(t) = EXP M(1-e-t ln2/Hs)
we can determine the time required to reach a half maximum size (h) as follows:
The h constant should be inversely proportional to the initial growth rate or
dD/dt = A (x / (x + B')) = K/Hs
Hs = K (x + B') / A x
Hs = K' (x + B') / x
Plant size with increasing amounts of nutrient according to Mitscherlich is
Y(x) = A(1 - e-x ln2/h)
If we let M(x) = lnY(x) then the Compertzian equation becomes
Y(t) = EXP M(x)(1-e-t ln2/Hs )
Now if we let h = 5 (the quantity of nutrient required to give a half maximum yield) and we let K' = 100, B' = 5, and A = 100 then from these values we can plot a series of time courses which will allow us to determine the theoretical yields at various times for various amounts of nutrient. From the Mitscherlich equation we know that if we add 2.5 grams of nutrient this amount should yield a maximum weight of 29.3 grams of plant weight, 5 grams should yield 50 grams of plant weight and so on up to 100 grams of biomass. These figures along with the number of effect factors (nh) are given in the following table:
Table 2
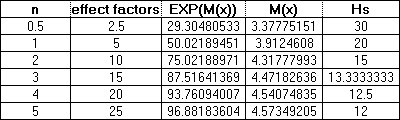
The values M(x) and Hs from the above table can now be used to determine the weight of a plant at various times after planting from the following formulas
Y(t) = EXP [3.38(1-e-(ln2/30) t)]
Y(t) = EXP [3.91(1-e-(ln2/20) t)]
Y(t) = EXP [4.32(1-e-(ln2/15) t)]
Y(t) = EXP [4.47(1-e-(ln2/13.33) t)]
Y(t) = EXP [4.54(1-e-(ln2/12.5) t)]
Y(t) = EXP [4.57(1-e-(ln2/12) t)]
Plots of the above equations are shown in figure 12.
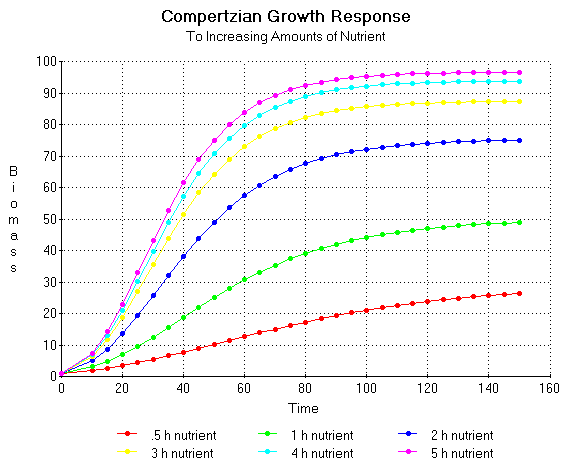
Figure 12. Hypothetical Compertzian growth trajectories of a plant given various quantities of nutrient expressed in Baule units (quantity of nutrient required to give a half maximal yield). The time required to reach a half the maximum mass (Hs) was determined using a derivation of the Rauterberg equation: Hs = K' (x + B') / x. The M constant was derived from the Mitscherlich equation: M(x) = A(1 - e-x ln2/h). These values were substituted into the Compertz equation: Y(t) = EXP M(x)(1-e-t ln2/h).
Now we can use slices from the above growth curve to get an idea of how the growth response of a plant might change with increasing amounts of nutrient at different points in the plant's vegetative life span. Figure 13 shows the growth response at different times with the theoretical points taken from Figure 12.
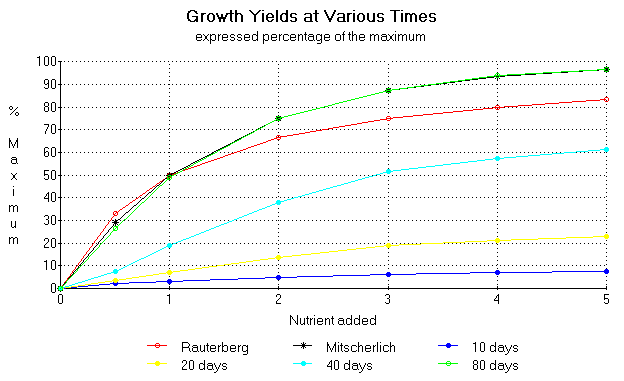
Figure 13. Theoretical nutrient growth response curves taken at different times during the vegetative life span of a plant assuming Compertzian growth dynamics. Data points taken from figure 12. Nutrient addition is expressed in effect factors.
We can see from the above figure that the growth curve starts as almost linear at 10 days, then gradually changes to a hyperbolic curve at 40 days and then finally becomes logarithmic at 80 days. The figure shows the Mitscherlich and Rauterberg curves for comparison.
If the growth trajectories are expressed as a fraction of the maximum with time as is seen in figure14 then the relationship between the yield of crops and the amount of nutrient becomes clearer. In this particular situation it is clear that one would have to wait at least 200 days before a proper analysis of the nutrient requirements can be made. Assumptions made on the growth requirements and nutrient interactions would be invalid if this procedure was not followed.
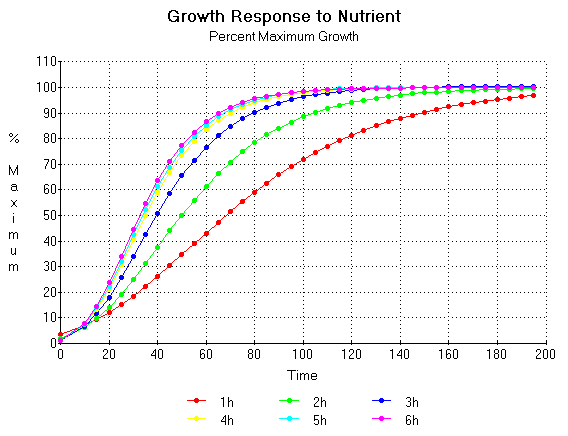
Figure 14. Growth trajectories from figure 12 expressed as a fraction of the maximum with time.
However, it can be seen from this form of the Compertz growth equation that the nutrient concentration at which growth rate is half maximal, is graphically coincident with the quantity of nutrient at which there is a half maximal yield.
Thus, with several growth rate data points it may be possible to fit curves which adequately project the maximum yield from the growth rate data alone. In other words the growth rate limiting nutrients are the same as the growth extent limiting nutrients.
Liebscher's Law of the Optimum Revisited
The hyperbolic equation does not adequately express the effect of nutrient concentration on the growth rate of plants in all situations, however. In some cases micronutrients may have toxic effects even at relatively low concentrations. Macronutrients, of course, can cause fertilizer "burn" by drawing out water from the plant causing it to wilt. Human cells grown in high concentrations of unfractionated serum become distorted in appearance and shrink probably for the same reason. In these situations the growth rate equation dD/dt = A [x] / ([x] + B) - C[x]
where C = a constant, may describe the relationship between growth rate and nutrient concentration more adequately .
Figure 15 shows the effect of the growth rate inhibitory constant, C, on the shape of the nutrient concentration response curve.
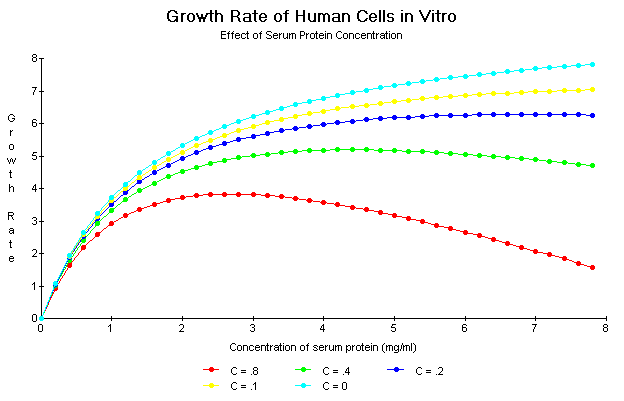
Figure 15. Effect of the C constant on the shape of the growth rate curve for various nutrient concentrations. As C decreases from 0.8 to 0 the shape of the curve becomes more and more hyperbolic.
The kinetics of the response where C does not equal zero shows an optimum growth rate. This explains many of the experimental results cited by DeVries and explains the basic principle behind Liebscher's law of the optimum. As in the case of Rauterburg(1939) studies which show a decline in use efficiency with an increase in a second nutrient are probably describing a growth rate response rather than a growth extent response.
The kinetics of Compertzian trajectories become quite complicated when the inhibitory effect of high nutrient concentrations are taken into account. Figure 16 shows the growth trajectories computed from the same equations as used for figure 12 except with different multiples of the effect factor h and with the m constant computed from the expression m= K/(A [x] / ([x] + B) - C[x]) with C = 0.0525.
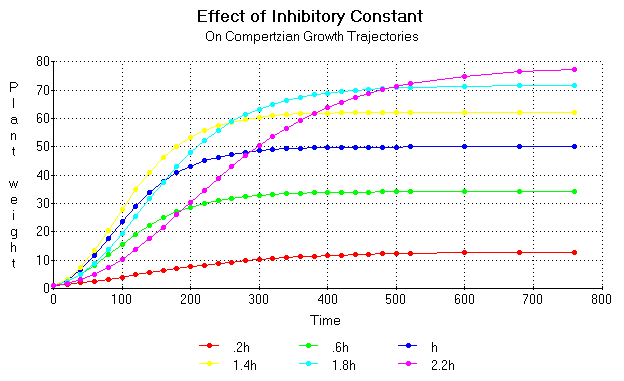
Figure 16. Growth trajectories computed from the same equations as used for figure 12 except with different multiples of the effect factor h and with the m constant computed from the expression m = K/(A [x] / ([x] + B) - C[x]).
Taking slices at different times from the above graph we can see how the computed use efficiency would change had we taken weight measurements at 120, 220, 320, 420, 520, and 1720 days in figure 17. It can be seen that with time Liebscher's Law of the Optimum becomes the Mitscherlich Law. That is, that the growth increment is proportional to the decrement from the maximum yield.
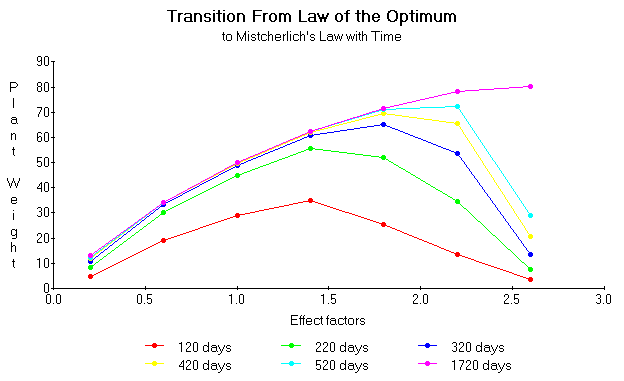
Figure 17. Growth trajectories computed from the same equations as used for figure 12 except with different multiples of the effect factor h and with the m constant computed from the expression m = K/(A [x] / ([x] + B) - C[x]).
Engelstad, O. P. and F. E. Khasawnch (1969) Use of a concurrent Mitscherlich model in fertilizer evalutation. Agron. J. 61: 473-475.
Macy, Paul (1935) The quantitative mineral nutrient requirements of plants. Plant Physiology :749-764.
Boss, Manley L. (1962) The Mitscherlich equation as applied to endogenous nutrient evaluation. Growth 26: 265-271.
McKeehan, W. L. and K. A. McKeehan (1981) Extracellular regulation of fibroblast multiplication: A direct kinetic approach to analysis of role of low molecular weight nutrients and serum growth factors. J. of supramol. Struct. 15: 83-110.
McKeehan, W. L., K. A. McKeehan and D. Calkin (1980) Extracellular Regulation of fibroblast multiplication. Quantitative differences in nutrient and serum factor requirements for multiplication of normal and SV40 virus- transformed human lung cells. J. Biol. Chem. 256: 2973- 2978.
Laird, Anna Kane, Tyler, S. A., and Barton, A. D. (1965) Dynamics of normal growth. Growth 29: 233-248.
Laird, Anna Kane (1965) Dynamics of relative growth. Growth 29: 249-263.
Niklas, Karl J. (1994) Plant allometry: the scaling of form and process Chicago : University of Chicago Press.
Mitscherlich, E.A. 1909. Des Gesetz des Minimums und das Gesetz des abnehmended Bodenertrages. Landwirsch. Jahrb. 3: 537-552.
Rauterberg, E. (1939) Uber die Beziehung zwischen Wachstumsfaktor und Ertrag unter resonderer Berucksichtigung der Berechnung der Konstanten A und c in der logarithmischen Gleichung von Mitscherlich. Bodenkunde und Pflanzenernahrung 54:10- 28.
Tisdale, S. L. (1975) Soil fertility and fertilizers. Macmillan Publishing Co., Inc. New York, New York.
Troughton, Arthur (1955) Studies on the growth of young grass plants with special reference to the relationship between the shoot and root systems. (find journal): 56- 65.
(1988) Growth and serum concentration concentration response curves of dermal fibroblasts from normal individuals and from individuals with adnenomatosis of the colon and rectum. Florida Atlantic University Master's thesis abstracts.
Vries, O. de (1939) Ertragskurven, Ertragsgesetze und die Bedeutung der Gegenseitigen Beeinflussung der Wachstumsfaktoren. Bodenkunde und Pflanzenernahrung 14: 1-10.